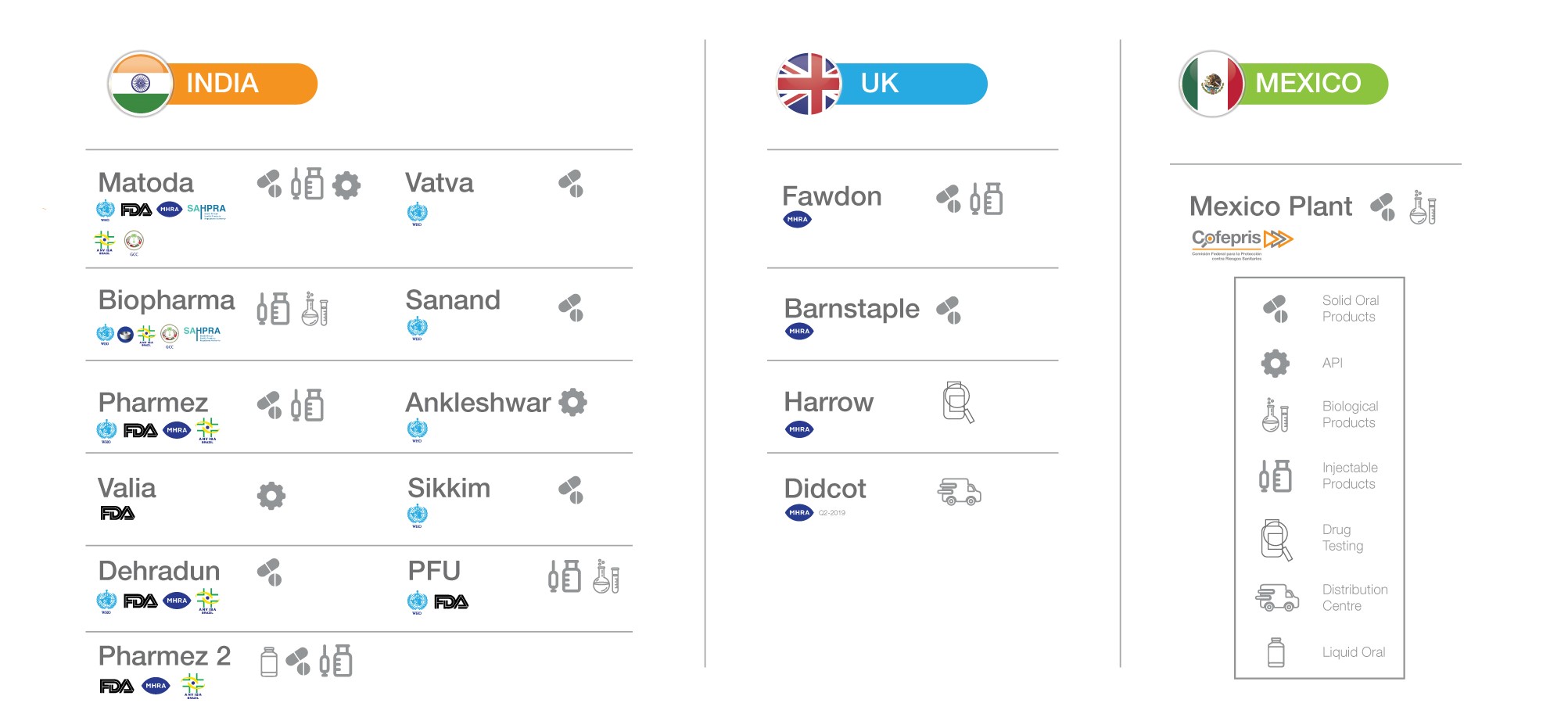
Facilities
Intas operates fourteen manufacturing facilities, of which eleven are located in India and the rest in UK and Mexico. The company also has an advanced analytical lab and state-of-the-art distribution centre in the UK. It operates two API and intermediate manufacturing facilities, each of which complies with the regulatory requirements in the jurisdictions in which it operates. Between them, these facilities have received approval from various prominent international regulatory bodies, including from U.S. Food and Drug Administration (FDA).
Each of Intas’ manufacturing facilities is designed, equipped and operated to deliver high-quality products within defined cost and delivery schedules. These manufacturing facilities have the flexibility to operate in various dosage forms and a wide range of batch sizes. Intas’ world class oncology formulation facility operates under global regulatory and safety standards. The Ahmedabad SEZ caters exclusively to the US, Europe and other regulated markets.
As a part of its progressive future strategy, Intas has invested in Pharmez 2 – a highly advanced expansion of its Pharmez SEZ facility in Gujarat, India. This new age, highly automated and efficient manufacturing layout has the capacity to produce more than one billion solid orals per month as well as 5 million injectable units. The futuristic Pharmez 2 facility is set to be the cornerstone for Intas’ promising future.
Manufacturing Facilities